Do I need an adult RSV vaccine?
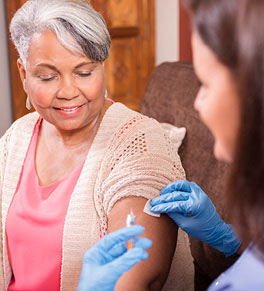
RSV — short for respiratory syncytial virus — may cause only mild cold-like symptoms for most people, but it can be especially severe for older adults.
Now two vaccines are approved to prevent RSV in adults age 60 and older.
“This an exciting new development,” says Dr. Hannah Nam, a UCI Health infectious disease specialist. “RSV is a highly contagious virus that can make it difficult to breathe and, in severe cases, could lead to hospitalization for pneumonia, other lung infections and heart failure exacerbation for older adults.”
Both vaccines are expected to be released later this year. She said patients should consult with their doctors to determine whether to get an RSV vaccine.
The U.S. Centers for Disease Control and Prevention estimates that complications of RSV infections are responsible for 60,000 to 160,000 hospitalizations and as many as 10,000 deaths each year in adults age 65 and older.
The two vaccines recently approved by the U.S. Food and Drug Administration (FDA) will be especially helpful for older residents in nursing homes and long-term care facilities, those living with young children who may be exposed to RSV in daycare centers and schools, as well as patients with health conditions that increase their risk for severe RSV infection.
These health conditions include:
- Chronic heart diseases such as congestive heart failure or coronary artery disease
- Chronic lung diseases such as asthma or chronic obstructive pulmonary disorder (COPD)
- Diabetes
- Liver and kidney disorders
- Hematologic disorders
- Neurologic disorders
- Having a compromised immune system, such as patients undergoing chemotherapy
Nam, who recently discussed the importance and safety of the new vaccines with VeryWell Health, shares her thoughts with Live Well.
How effective are the vaccines?
Both vaccines give up to two years of protection and are expected to be released for use later this year.
The Pfizer vaccine, Abrysvo, was 86% effective against RSV cases in study participants who had three or more symptoms within the first year. When looked at for two consecutive seasons, the efficacy was still 79% against the virus.
The GSK vaccine, Arexvy, was reported to be nearly 83% effective against RSV and prevented hospitalization for 94.1% of participants. During the second season, its ability to prevent symptomatic RSV infection decreased to 56.1% of those studied but still prevented hospitalizations in 64.2% of participants.
What type of vaccines are they?
Both vaccines are protein subunit vaccines, which use a harmless version of a protein found on the virus to stimulate the body’s immune system. This type of vaccine technology is already well known to us.
Neither vaccine contains any live virus so they cannot give you the RSV infection.
Arexvy also contains an adjuvant, an ingredient that also boosts the immune system. Vaccines that use a similar technology are the pneumococcal vaccine (Pneumovax or Prevnar), the shingles vaccine (Shingrix) and the hepatitis B vaccine (Heplisav-B).
Are they safe and what are the side effects?
Overall, studies have shown that the RSV vaccines are safe and have been very well tolerated. The most common side effects are similar to other vaccines, including pain at the injection site, fatigue, muscle pain, headache and joint stiffness.
The Arexvy (GSK) vaccine was tested on 12,500 people. There were 10 cases of atrial fibrillation (i.e., irregular heart rhythm) among vaccine recipients within a month of vaccination, compared with four in the control group.
There were also a few autoimmune syndromes observed among Arexvy recipients. Two patients who received the influenza and Arexvy RSV vaccines at the same time developed a neurological disorder called acute disseminated encephalomyelitis (ADEM). It is not known if either or both vaccines were the cause.
One patient who received the RSV vaccine developed Guillain-Barré syndrome, a rare neurological disorder that causes the immune system to attack the body’s peripheral nervous system. It is unclear whether this is a significant increase over the background incidence of Guillain-Barré syndrome that already occurs in older adults.
Among the 20,255 people who received Abryvso (Pfizer), one case of Guillain-Barré syndrome and one case of another rare neurological disorder called Miller Fisher syndrome were reported.
While these cases were rare events and occurrences have happened with other vaccines and infections (including RSV, itself), the FDA has asked the companies to study it further.
When should I get the RSV vaccine?
Because a single dose is thought to given protection for at least two years, it is recommended that older adults get an RSV vaccine as soon as they are available.
They are most effective when given before RSV season, which is typically from October to May.
There are further studies being done to determine whether booster doses may be needed.
Can I get the vaccine with my regular shots?
The CDC says RSV vaccines can be given at the same time as other vaccines, including influenza.
With COVID-19, influenza and RSV viruses often circulating at the same time, getting all three vaccines can significantly reduce the threat of serious illness and hospitalization.
Any final recommendations?
It is very common for there to be side effects for any vaccine, including rare ones like Guillain-Barré. The FDA continuously monitors and collects data on all vaccine side effects and this is being done with the new RSV vaccines.
These vaccines are considered safe and effective in preventing severe illness, hospitalization and death from RSV in older adults.
So, if you or someone you love is over 60 years of age, ask your primary care physician whether one of these vaccines is right for you.
Related stories
Explore further
Browse more blog posts by topic.




