A novel approach to treating knee arthritis
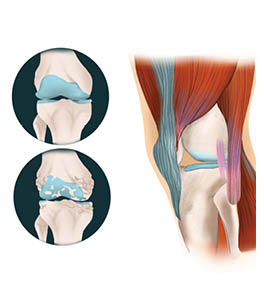
Cartilage is a shock absorber in the knee joint, but age, injury and inflammation can erode it, as shown in the examples on the left. An investigational intramuscular injection may help strengthen the muscles around the knee. "I don't know of any other treatments that target the muscle," UCI orthopedic surgeon Dr. Dean Wang says. "That’s what is novel about this therapy.”
About 13% of women and 10% of men ages 60 or older have knee osteoarthritis, a leading cause of disability among Americans. Many of them undergo knee replacement surgery to retain mobility.
But what if there was a way to strengthen the muscles around the knee to alleviate pain and preserve knee function without a joint replacement? To find out, UCI Health researchers are conducting the first-in-human clinical trial of a stem cell-derived treatment that could help keep aging knees strong and pain-free.
Sponsored by Immunis Inc., the IMM01-STEM study tests an injectable solution that targets muscle atrophy in patients with mild to moderate knee osteoarthritis, says orthopedic surgeon Dr. Dean Wang, who is leading the novel trial at the UCI Sue and Bill Gross Stem Cell Research Center’s Alpha Clinic.
By strengthening the leg muscles surrounding the knee, which helps reduce the pain, the investigational treatment developed by the Irvine-based biotech company has the potential to become an alternative to other therapies, including joint replacement, for the degenerative condition that often strikes physically active people in their 50s and 60s, says Wang.
Wear and tear on cartilage
In osteoarthritis, knee cartilage — the strong, flexible connective tissue that acts as a shock absorber within the joint — gradually wears down with age, inflammation or other factors. Doctors have limited options for treating the condition, says Wang, who specializes in orthopedic sports medicine and joint preservation surgery for knees and hips as well as shoulders and elbows.
But patients under age 60 or those whose osteoarthritis isn’t severe enough usually aren’t considered ideal candidates for knee replacement because the artificial implants last only about 15 years. Doctors instead may prescribe anti-inflammatory medications and physical therapy.
There are newer treatments, such as platelet-rich plasma injections, that have shown some success, but Wang says it is difficult to predict who will benefit from those therapies.
That leaves otherwise healthy middle-aged people with few alternatives.
“Most of these patients experience some muscle-related atrophy because they’ve been in pain for a while and can’t really exercise to build their strength,” he says.
A novel alternative
The IMMO1-STEM treatment could be a powerful remedy to reverse muscle atrophy and prevent knee arthritis from worsening.
The investigational therapy is an injectable solution created by stimulating human stem cells to produce a set of proteins called a secretome.
In preclinical research with animals, Immunis scientists showed that these proteins not only suppressed cellular wasting processes, they also improved muscle mass and strength — an effect comparable to the way muscles grow from regular workouts.
The therapy also affords greater safety “because we’re not injecting stem cells themselves but rather the growth factors they produce,” Wang explains. “Anytime you inject foreign cells, an immune reaction is possible.”
Safer and less invasive
Intramuscular injections are also less invasive and painful than other therapeutic options, he says.
“Most investigational and standard-of-care treatments are injected inside the joint itself. I don’t know of any other treatments that target the muscle. That’s what is novel about this therapy.”
To be eligible for the phase 1/2a trial, participants must have tried at least one standard treatment for knee osteoarthritis without success. Up to 18 patients ages 50 to 75 will receive twice-weekly injections of the compound over a four-week period. Leg strength will be measured before and after the injection regimen, and MRI scans will assess changes in the knee muscles.
If the initial research shows the therapy is safe, additional trials will be needed to determine its effectiveness.
Wang is hopeful that this treatment will improve function and decrease his patients’ pain.
“We have a lot of 50-year-olds who want to be active, who want to bike, surf and run. Right now, these are probably some of my most difficult patients to treat. This could be a big help to them.”
Immunis Inc. is a private biotechnology company that is developing a novel immunomodulatory secretome product for various manifestations of age and disease-related immune decline. The STEM product line leverages Immunis’ leading-edge capabilities in stem cell technologies to deliver a product of all natural, all human immune modulators in their natural relative physiological concentrations. The biotech firm has deep ties to UC Irvine and neuroscience research. Its chairman is Hans Keirstead, PhD, an internationally known stem cell expert and former UCI professor who helped raise funds for and was co-director of the $77 million stem cell research center. Its chief science officer is Thomas Lane, PhD, who directs the UCI Center for Virus Research and is a Chancellor's Professor of Neurobiology & Behavior.
The UCI Alpha Clinic is the clinical trial arm of the UCI Sue & Bill Gross Stem Cell Research Center and part of a network of the leading medical centers funded by the California Institute for Regenerative Medicine. It specializes in delivering leading-edge stem cell clinical trials and gene therapy to patients, and it seeks to accelerate the development of new treatments through partnerships with patients, medical providers and clinical trial sponsors. Visit stemcell.uci.edu to learn more about regenerative medicine research at UCI.
Explore further
Browse more blog posts by topic.




